More
From Silicon to Synthesis: A Beginner’s Guide to Computer-Aided Drug Design (CADD)
29 Apr , 2026
The days of "spray and pray" drug discovery are over. Traditionally, identifying a single "hit" compound required the physical screening of millions of chemicals—a process that costs millions of dollars and months of laboratory time.
As we progress through 2026, Computer-Aided Drug Design (CADD) has transitioned from a supporting tool to the primary driver of pharmaceutical R&D. If you aren't using computational modeling before you pick up a pipette, you’re already behind.
The CADD Advantage: Efficiency by Design
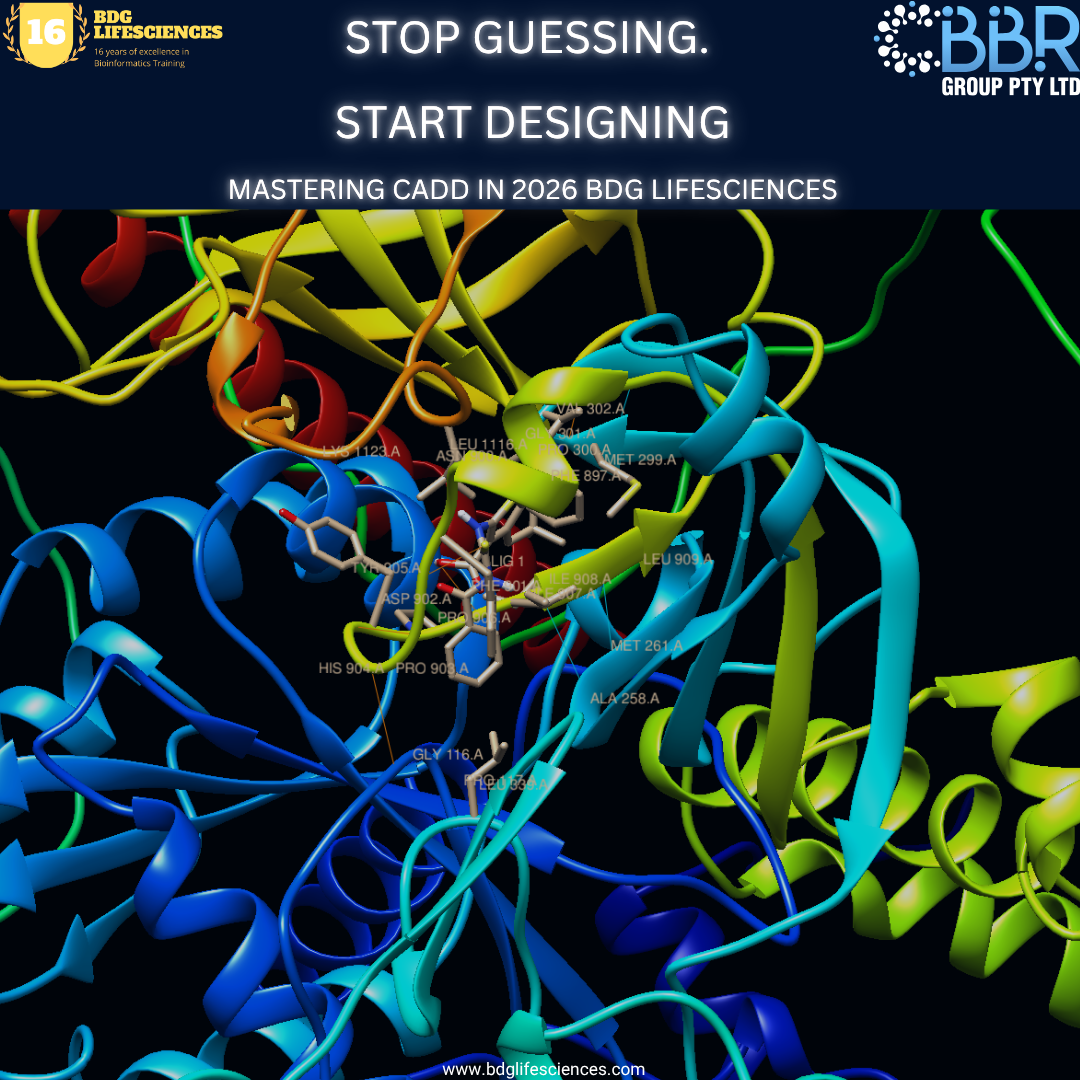
CADD allows researchers to virtually "dock" millions of potential drugs into a target protein’s binding pocket. It’s not just about speed; it’s about rationality. Instead of guessing which molecules might work, we use physics and AI to predict which ones will work.
The Two Pillars of Modern CADD:
1. Structure-Based Drug Design (SBDD): When the 3D structure of a target (like a viral protein) is known, we use SBDD. With the recent release of AlphaFold 3 and enhanced Molecular Dynamics (MD) simulations, we can now see how a drug behaves in a moving, "breathing" biological system—not just a static image.
2. Ligand-Based Drug Design (LBDD): If the target’s structure is a mystery, we look at known successful drugs (ligands) and use Pharmacophore Mapping. We find the "common denominator" that makes those drugs work and design new, more potent molecules based on that blueprint.
Breaking the Synthesis Bottleneck
One of the biggest trends this year is the integration of Retrosynthesis AI. It’s one thing to design a "miracle molecule" on a computer; it’s another to actually build it. Modern CADD tools now tell us exactly how to synthesize the molecule in the lab, ensuring that our virtual designs are commercially and chemically viable.
Why BDG LifeSciences?
The barrier to entry for CADD used to be high-performance computing (HPC) costs. Today, cloud-native platforms have democratized this. At BDG, we empower the next generation of researchers to master tools like AutoDock Vina, GROMACS, and PyMOL, bridging the gap between computational theory and real-world therapeutic results.




